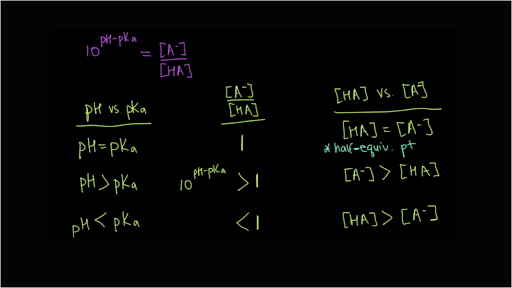
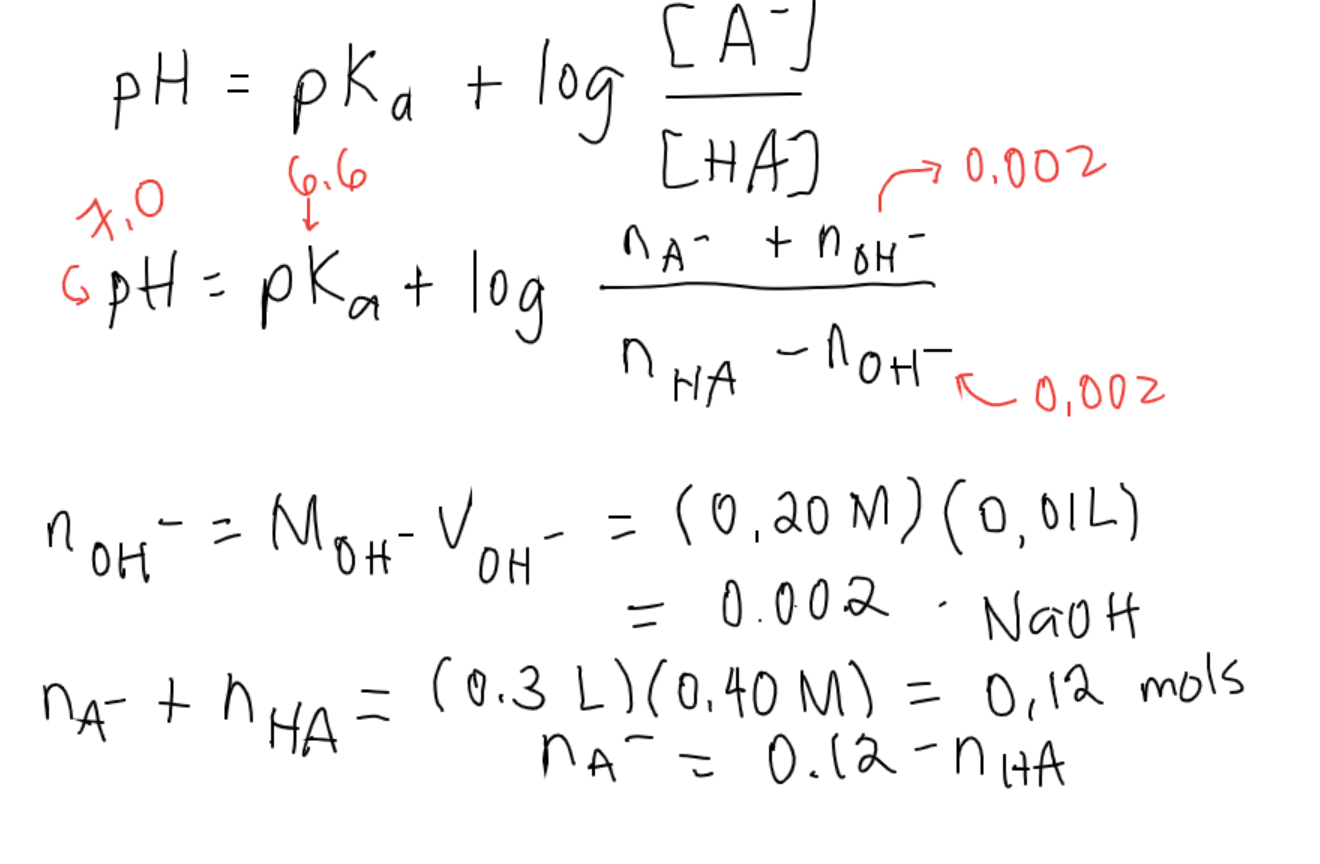![SOLVED: What is the pH of a buffer composed of 0.10 M oxalic acid and 0.08 M oxalate ion? The pKa for oxalic acid is 1.23. pH = pKa log ([A-J[HA]) Use SOLVED: What is the pH of a buffer composed of 0.10 M oxalic acid and 0.08 M oxalate ion? The pKa for oxalic acid is 1.23. pH = pKa log ([A-J[HA]) Use](https://cdn.numerade.com/ask_images/08a0de4c43b94a8e8f10c82ca0bf2b68.jpg)
SOLVED: What is the pH of a buffer composed of 0.10 M oxalic acid and 0.08 M oxalate ion? The pKa for oxalic acid is 1.23. pH = pKa log ([A-J[HA]) Use

pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems - YouTube

How should the acid dissociation constant pKa be measured? | Automatic Potentiometric Titrators | Faq | Kyoto Electronics Manufacturing Co.,Ltd.("KEM")

SOLVED: For an acidic drug, the following relationship between logD and lggE exists when pH is much greater than pKa of the drug: LogD = LogP (pH pKa) Derive the above equation

A plot of the pH dependence of the solubility of an acid with pKa 4.8... | Download Scientific Diagram

pKa & pH Values| Functional Groups, Acidity & Base Structures - Video & Lesson Transcript | Study.com
![Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube](https://i.ytimg.com/vi/SLPu7qlUdEA/maxresdefault.jpg)


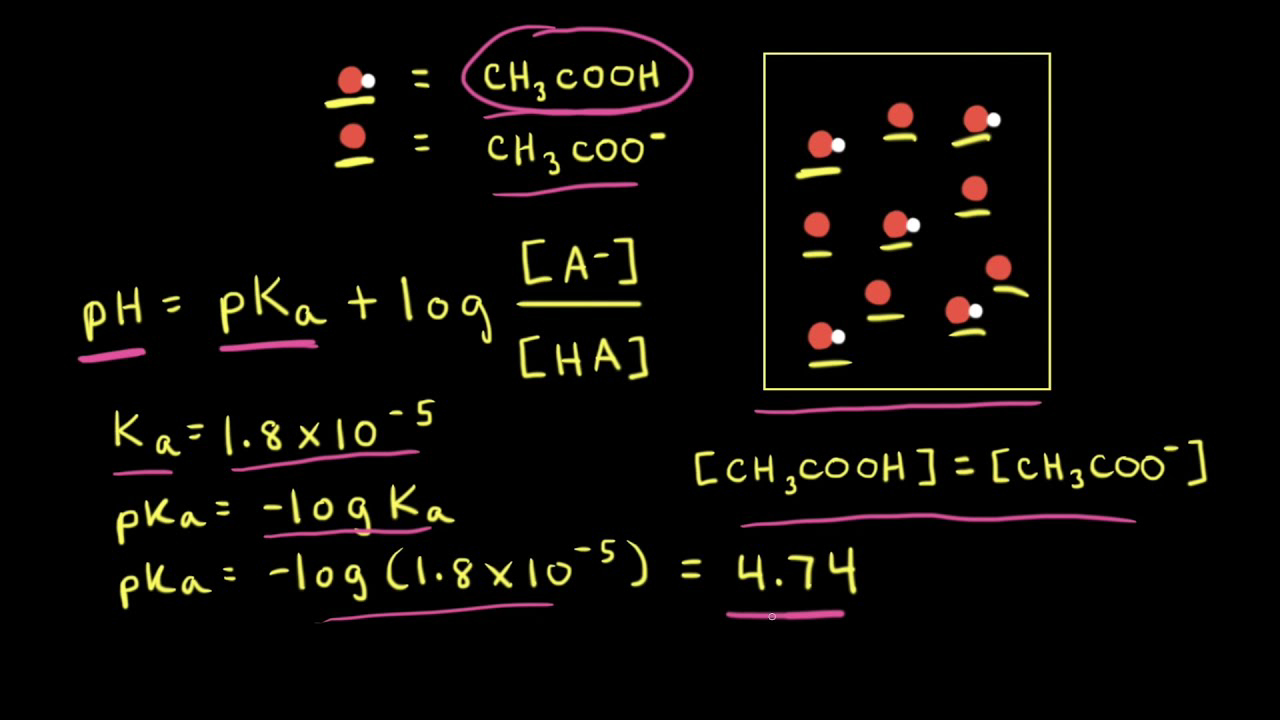


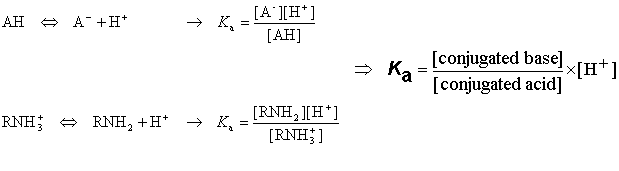


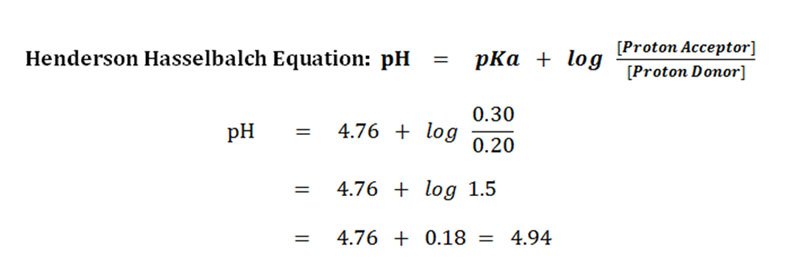
:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)